Abstract
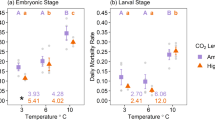
This study investigated the effects of seawater pH (i.e., 8.10, 7.85 and 7.60) and temperature (16 and 19 °C) on (a) the abiotic conditions in the fluid surrounding the embryo (viz. the perivitelline fluid), (b) growth, development and (c) cuttlebone calcification of embryonic and juvenile stages of the cephalopod Sepia officinalis. Egg swelling increased in response to acidification or warming, leading to an increase in egg surface while the interactive effects suggested a limited plasticity of the swelling modulation. Embryos experienced elevated pCO2 conditions in the perivitelline fluid (>3-fold higher pCO2 than that of ambient seawater), rendering the medium under-saturated even under ambient conditions. The growth of both embryos and juveniles was unaffected by pH, whereas 45Ca incorporation in cuttlebone increased significantly with decreasing pH at both temperatures. This phenomenon of hypercalcification is limited to only a number of animals but does not guarantee functional performance and calls for better mechanistic understanding of calcification processes.





Similar content being viewed by others
References
Addadi L, Joester D, Nudelman F, Weiner S (2006) Mollusk shell formation: a source of new concepts for understanding biomineralization processes. Chem Eur J 12:980–987. doi:10.1002/chem.200500980
Appellöf A (1893) Die Schalen von sepia, spirula, and nautilus. Studien über den Bau und das Wachstum. Kongl Svenska Vetensk Acad Handl 25:1–106. ISSN 0023-5377
Barbieri E, Barry K, Child A, Wainwright N (1997) Antimicrobial activity in the microbial community of the accessory nidamental gland and egg cases of Loligo pealei (Cephalopoda: Loliginidae). Biol Bull 193(2):275–276
Bégovic M, Copin-Montégut C (2002) Processes controlling annual variations in the partial pressure of CO2 in surface waters of the central northwestern Mediterranean Sea (Dyfamed site). Deep-Sea Res PT II 49:2031–2047
Beniash E, Aizenberg J, Addadi L, Weiner S (1997) Amorphous calcium carbonate transforms into calcite during sea urchin larval spicule growth. Proc Roy Soc B 264:461–465. doi:10.1098/rspb.1997.0066
Beniash E, Addadi L, Weiner S (1999) Cellular control over spicule formation in sea urchin embryos: a structural approach. J Struct Biol 125:50–62. doi:10.1006/jsbi.1998.4081
Bijma J, Honisch B, Zeebe RE (2002) The impact of the ocean carbonate chemistry on living foraminiferal shell weight: comment on ‘‘Carbonate ion concentration in glacial-age deep waters of the Carribbean Sea’’ by W. S. Broecker and E. Clark. Geochem Geophys Geosyst 3(11):1064. doi:10.1029/2002GC000388
Birchall JD, Thomas NL (1983) On the architecture and function of cuttlefish bone. J Mater Sci 18:2081–2086. doi:10.1007/BF00555001
Blackford JC (2010) Predicting the impacts of ocean acidification: challenges from an ecosystem perspective. J Marine Syst 81(1–2):12–18. doi:10.1016/j.jmarsys.2009.12.016
Boletzky SV (1974) The “larvae” of Cephalopoda: a review. Thalassia Jugoslavica 10:45–76
Boletzky SV (1983) Sepia officinalis cephalopod life cycles species account. In: Boyle PR (ed) Cephalopod life cycle. Academic Press, London, pp 31–52
Boletzky SV (1986) Encapsulation of cephalopod embryos: a search for functional correlations. Am Malacol Bull 4:217–227
Boletzky SV (1989) Recent studies on spawning, embryonic development, and hatching in the cephalopoda. Adv Mar Biol 6:86–109
Boletzky SV (2003) Biology of early life stages in cephalopod molluscs. Adv in Mar Biol 44:143–203. doi:10.1016/S0065-2881(03)44003-0
Bustamante P, Teyssié JL, Fowler SW, Cotret O, Danis B, Miramand P, Warnau M (2002) Biokinetics of zinc and cadmium accumulation and depuration at different stages in the life cycle of the cuttlefish Sepia officinalis. Mar Ecol Prog Ser 231:167–177
Bustamante P, Teyssié J, Danis B, Fowler SW, Miramand P, Cotret O (2004) Uptake, transfer and distribution of silver and cobalt in tissues of the common cuttlefish Sepia officinalis at different stages of its life cycle. Mar Ecol Prog Ser 269:185–195. doi:10.3354/meps269185
Bustamante P, Teyssié J-L, Fowler SW, Warnau M (2006) Contrasting bioaccumulation and transport behaviour of two artificial radionuclides (241Am and 134Cs) in cuttlefish eggshell. Vie Milieu 56(2):153–156
Byrne M (2011) Impact of ocean warming and ocean acidification on marine invertebrate life history stages: vulnerabilities and potential persistence in a changing ocean. Oceanogr Mar Biol Annu Rev 49:1–42
Checkley DM, Dickson AG, Takahashi M, Radich JA, Eisenkolb N, Asch R (2009) Elevated CO2 enhances otolith growth in young fish. Science 324:1683. doi:101126/science1169806S
Comeau S, Gattuso J-P, Nisumaa A-M, Orr J (2011) Impact of aragonite saturation state changes on migratory pteropods. Proc Roy Soc B. doi:10.1098/rspb.2011.0910
Craig S, Overnell J (2003) Metals in squid, Loligo forbesi, adults, eggs and hatchlings. No evidence for a role for Cu- or Zn-metallothionein. Comp Biochem Phys C 134(3):311-317. doi:10.1016/S1532-0456(02)00274-0
Cronin ER, Seymour RS (2000) Respiration of the eggs of the giant cuttlefish Sepia apama. Mar Biol 136:863–870. doi:10.1007/s002270000274
Denton EJ, Gilpin-Brown JB (1959) Buoyancy of the cuttlefish. Nature 184:1330–1331. doi:10.1038/1841330a0
Denton EJ, Gilpin-Brown JB (1961) The buoyancy of the cuttlefish, Sepia officinalis (L.). J Mar Biol Assoc UK 41:319–342. doi:10.1017/S0025315400023948
Denton EJ, Taylor DW (1964) The composition of gas in the chambers of the cuttlebone of Sepia officinalis. J Mar Biol Assoc UK 44:203–207. doi:10.1017/S0025315400024747
Denton EJ, Gilpin-Brown JB, Howarth JV (1961) The osmotic mechanism of the cuttlebone. J Mar Biol Assoc UK 41:351–363. doi:10.1017/S0025315400023961
Dickson AG, Millero F (1987) A comparison of the equilibrium constants for the dissociation of carbonic acid in seawater media. Deep-Sea Res 34:1733–1743. doi:10.1016/0198-0149(87)90021-5
Dickson AG, Sabine CL, Christian JR (2007) Guide to best practices for ocean CO2 measurements. PICES Spec Publ 3
Dupont S, Thorndyke MC (2009) Impact of CO2-driven ocean acidification on invertebrates early life-history—what we know, what we need to know and what we can do? Biogeosci Discussions 6:3109–3131. doi:10.5194/bgd-6-3109-2009
Dupont S, Ortega-Martínez O, Thorndyke MC (2010) Impact of near-future ocean acidification on echinoderms. Ecotoxicology 19:449–462. doi:10.1007/s10646-010-0463-6
Dupont S, Dorey N, Stumpp M, Melzner F, Thorndyke M (2012) Long term and trans life-cycle effects of exposure to ocean acidification in the green sea urchin Strongylocentrotus droebachiensis. Mar Biol. doi:10.1007/s00227-012-1921-x
Fabry VJ, Balch WM (2010) Direct measurements of calcification rates in planktonic organisms. In: Riebesell U, Fabry VJ, Hanson L, Gattuso JP (eds) Guide for best practices in ocean acidification research and data reporting. Publications Office of the European Unit, Luxembourg, pp 201–212
Findlay HS, Wood HL, Kendall MA, Spicer JI, Twitchett RJ, Widdicombe S (2011) Comparing the impact of high CO2 on calcium carbonate structures in different marine organisms. Mar Biol Res 7:565–575
Forsythe JW, Lee P, Walsh L, Clark T (2002) The effects of crowding on growth of the European cuttlefish, Sepia officinalis (Linnaeus, 1758) reared at two temperatures. J Exp Mar Biol Ecol 269(2):173–185. doi:10.1016/S0022-0981(02)00006-0
Gazeau F, Gattuso JP, Dawber C, Pronker AE, Peene F, Peene J, Heip CHR, Middelburg JJ (2010) Effect of ocean acidification on the early life stages of the blue mussel Mytilus edulis. Biogeosciences 7:2051–2060. doi:10.5194/bg-7-2051-2010
Gazeau F, Gattuso J-P, Greaves M, Elderfield H, Peene J, Heip CHR, Middelburg JJ (2011) Effect of carbonate chemistry alteration on the early embryonic development of the Pacific oyster (Crassostrea gigas). PLoS ONE 6:e23010. doi:10.1371/journal.pone.0023010
Gomi F, Masamich Y, Nakazawa T (1986) Swelling of egg during development of the cuttlefish, Sepiella japonica. Zool Sci 3(4):641–645
Gooding RA, Harley CDG, Tang E (2009) Elevated water temperature and carbon dioxide concentration increase the growth of a keystone echinoderm. P Natl Acad Sci USA 106:9316–9321
Guerra A (2006) Ecology of Sepia officinalis. Vie milieu 56(2):97–107
Gutowska MA, Melzner F (2009) Abiotic conditions in cephalopod (Sepia officinalis) eggs: embryonic development at low pH and high pCO2. Mar Biol 156:515–519. doi:10.1007/s00227-008-1096-7
Gutowska MA, Pörtner H-O, Melzner F (2008) Growth and calcification in the cephalopod Sepia officinalis under elevated seawater pCO2. Mar Ecol Prog Ser 373:303–309. doi:10.3354/meps07782
Gutowska MA, Melzner F, Langenbuch M, Bock C, Claireaux G, Pörtner HO (2010a) Acid-base regulatory ability of the cephalopod (Sepia officinalis) in response to environmental hypercapnia. J Comp Physiol B 180:323–335. doi:10.1007/s00360-009-0412-y
Gutowska MA, Melzner F, Pörtner H-O, Meier S (2010b) Cuttlebone calcification increases during exposure to elevated seawater pCO2 in the cephalopod Sepia officinalis. Mar Biol 157:1653–1663. doi:10.1007/s00227-010-1438-0
Hoegh-Guldberg O, Mumby PJ, Hoote AJ et al (2007) Coral reefs under rapid climate change and ocean acidification. Science 318:1737–1742. doi:10.1126/science.1152509
Hu MY, Sucré E, Charmantier-Daures M, Charmantier G, Lucassen M, Himmerkus N, Melzner F (2010) Localization of ion-regulatory epithelia in embryos and hatchlings of two cephalopods. Cell Tissue Res 339:571–583. doi:10.1007/s00441-009-0921-8
Hu MY, Tseng Y-C, Stumpp M, Gutowska MA, Kiko R, Lucassen M, Melzner F (2011a) Elevated seawater pCO2 differentially affects branchial acid-base transporters over the course of development in the cephalopod Sepia officinalis. Am J Physiol 300:R1100–R1114. doi:10.1152/ajpregu.00653.2010
Hu MY, Tseng Y-C, Lin L-Y, Chen P-Y, Charmantier-Daures M, Hwang P–P, Melzner F (2011b) New insights into ion regulation of cephalopod molluscs: a role of epidermal ionocytes in acid-base regulation during embryogenesis. Am J Physiol 301:R1700–R1709. doi:10.1152/ajpregu.00107.2011
IPCC (2007) Fourth assessment report of the intergovernmental panel on climate change. In: Solomon, S, Qin D, Manning M, Chen Z, Marquis M, Averyt KB, Tignor M, Miller HL (eds) Climate change 2007. Cambridge University Press, Cambridge, United Kingdom and New York, NY
Jacob DE, Wirth R, Soldati AL, Wehrmeister U, Schreiber A (2011) Amorphous calcium carbonate in the shells of adult Unionoida. J Struct Biol 173:241–249. doi:10.1016/j.jsb.2010.09.011
Kleypas JA, Feely RA, Fabry VJ, Langdon C, Sabine CL, Robbins LL (2006) Impacts of ocean acidification on coral reefs and other marine calcifiers: a guide for future research, report of a workshop held 18–20 April 2005, St Petersburg, FL, sponsored by NSF, NOAA, and the US Geological Survey http://www.ucar.edu/communications/Final_acidification.pdf
Kroeker KJ, Kordas RL, Crim RN, Singh GG (2010) Review and synthesis: meta-analysis reveals negative yet variable effects of ocean acidification on marine organisms. Ecol Lett 13:1419–1434. doi:10.1111/j.1461-0248.2010.01518.x
Kurihara H (2008) Effects of CO2-driven ocean acidification on the early developmental stages of invertebrates. Mar Ecol Prog Ser 373:275–284. doi:10.3354/meps07802
Lacoue-Labarthe T, Warnau M, Oberhänsli F, Teyssié J-L, Koueta N, Bustamante P (2008) Differential bioaccumulation behaviour of Ag and Cd during the early development of the cuttlefish Sepia officinalis. Aquat Toxicol 86:437–446. doi:10.1016/j.aquatox.2007.12.005
Lacoue-Labarthe T, Martin S, Oberhänsli F, Teyssié J-L, Markich S, Jeffree R, Bustamante P (2009) Effects of increased pCO2 and temperature on trace element (Ag, Cd and Zn) bioaccumulation in the eggs of the common cuttlefish, Sepia officinalis. Biogeosciences 6:2561–2573. doi:10.5194/bg-6-2561-2009
Lacoue-Labarthe T, Warnau M, Oberhänsli F, Teyssié JL, Bustamante P (2010a) Contrasting accumulation biokinetics and distribution of 241Am, Co, Cs, Mn and Zn during the whole development time of the eggs of the common cuttlefish, Sepia officinalis. J Exp Mar Biol Ecol 382:131–138. doi:10.1016/j.jembe.2009.10.008
Lacoue-Labarthe T, Le Bihan E, Borg D, Koueta N, Bustamante P (2010b) Variation of acid phosphatases and cathepsins activities in the cuttlefish (Sepia officinalis) eggs: specific activity and effects of Ag, Cd, Cu exposures. ICES J Marine Sci 67:1517–1523. doi:10.1093/icesjms/fsq044
Lacoue-Labarthe T, Villanueva R, Rouleau C, Oberhänsli F, Teyssié J-L, Jeffree R, Bustamante P (2011a) Radioisotopes demonstrate the contrasting bioaccumulation capacities of heavy metals in embryonic stages of cephalopod species. PLoS ONE 6(11):e27653. doi:10.1371/journal.pone.0027653
Lacoue-Labarthe T, Réveillac E, Oberhänsli F, Teyssié JL, Jeffree R, Gattuso JP (2011b) Effects of ocean acidification on trace element accumulation in the early-life stages of squid Loligo vulgaris. Aquat Toxicol 105:166–176. doi:10.1016/j.aquatox.2011.05.021
Langdon C, Atkinson MJ (2005) Effect of elevated pCO2 on photosynthesis and calcification of corals and interactions with seasonal change in temperature/irradiance and nutrient enrichment. J Geophys Res 110:C09S07. doi:10.1029/2004JC002576
Lavigne H, Gattuso J-P (2009) Seacarb: seawater carbonate chemistry with R. R package version 2.4. http://CRAN.R-project.org/package=seacarb
Leersnyder M De, Lemaire J (1972) Sur la composition minérale du liquide périembryonnaire de l’oeuf de Sepia officinalis L. Cah Biol Mar 13:429–443
Lemaire J (1970) Table de développement embryonnaire de Sepia officinalis L (Mollusque Céphalopode). B Soc Zool Fr 95(4):773–782
Levitus S, Antonov J, Boyer T (2005) Warming of the world ocean, 1955–2003. Geophys Res Lett 32:L02604. doi:10.1029/2004GL021592
Lewis E, Wallace DWR (1998) Program developed for CO2 system calculations. ORNL/CDIAC-105. US Department of Energy, Oak Ridge, Tennessee. http://cdiac.ornl.gov
Martin S, Richier S, Pedrotti M-L, Dupont S, Castejon C, Gerakis Y, Kerros M-E, Oberhänsli F, Teyssié J-L, Jeffree R, Gattuso J-P (2011) Early development and molecular plasticity in the Mediterranean sea urchin Paracentrotus lividus exposed to CO2-driven acidification. The J Exp Biol 214:1357–1368. doi:10.1242/jeb.051169
Mehrbach C, Culberson CH, Hawley JE, Pytkowicz RM (1973) Measurement of the apparent dissociation constants of carbonic acid in seawater at atmospheric pressure. Limnol Oceanogr 18(6):897–907
Melzner F, Forsythe J, Lee P, Wood J, Piatowski U, Clemmesen C (2005) Estimating recent growth in the cuttlefish: are nucleic acid-based indicators for growth and condition the method of choice? J Exp Mar Biol Ecol 317:37–51. doi:10.1016/j.jembe.2004.11.011
Melzner F, Bock C, Pörtner H-O (2006) Temperature-dependent oxygen extraction from the ventilatory current and the costs of ventilation in the cephalopod Sepia officinalis. J Comp Physiol B 176:607–621. doi:10.1007/s00360-006-0084-9
Melzner F, Bock C, Pörtner H-O (2007) Allometry of thermal limitation in the cephalopod Sepia officinalis. Comp Biochem Phys A 146:149–154. doi:10.1016/j.cbpa.2006.07.023
Melzner F, Gutowska MA, Langenbuch M, Dupont S, Lucassen M, Thorndyke MC, Bleich M, Pörtner H-O (2009) Physiological basis for high CO2 tolerance in marine ectothermic animals: pre-adaptation through lifestyle and ontogeny? Biogeosciences 6:2313–2331. doi:10.5194/bg-6-2313-2009
Miller WA, Reynolds AC, Sobrino C, Riedel GF (2009) Shellfish face uncertain future in high CO2 world: influence of acidification on oyster larvae calcification and growth in estuaries. PLoS ONE 4:e5661. doi:10.1371/journal.pone.0005661
Miramand P, Bustamante P, Bentley D, Koueta N (2006) Variation of heavy metal concentrations (Ag, Cd, Co., Cu, Fe, Pb, V, Zn) during the life cycle of the common cuttlefish Sepia officinalis. Sci Total Environ 361:132–143. doi:10.1016/j.scitotenv.2005.10.018
Munday PL, Hernaman V, Dixson DL, Thorrold SR (2011) Effect of ocean acidification on otolith development in larvae of a tropical marine fish. Biogeosciences 8:1631–1641. doi:10.5194/bg-8-1631-2011
Orr J, Fabry VJ, Aumont O, Bopp L, Doney SC, Feely RA et al (2005) Anthropogenic ocean acidification over the twenty-first century and its impact on calcifying organisms. Nature 437:681–686. doi:10.1038/nature04095
Palmer AR (1992) Calcification in marine molluscs: how costly is it? P Natl Acad Sci USA 89:1379–1382. doi:10.1073/pnas.89.4.1379
Parker LM, Ross PM, O’Connor WA, Borysko L, Raftos DA, Pörtner H-O (2012) Adult exposure influences offspring response to ocean acidification in oysters. Glob Change Biol 18:82–92. doi:10.1111/j.1365-2486.2011.02520.x
Pechenik JA (2006) Larval experience and latent effects: metamorphosis is not a new beginning. Integr Comp Biol 46:323–333. doi:10.1093/icb/icj028
Pierce GJ, Allcock L, Bruno I et al. (2010) Cephalopod biology and fisheries in Europe. ICES cooperative research report, 303. ICES, Copenhagen, Denmark. ISBN 978-87-7482-078-9. http://oceanrep.geomar.de/10074/
Politi Y, Arad T, Klein E, Weiner S, Addadi L (2004) Sea urchin spine calcite forms via a transient amorphous calcium carbonate phase. Science 306:1161–1164. doi:10.1126/science.1102289
Pörtner H-O (2008) Ecosystem effects of ocean acidification in times of ocean warming: a physiologist’s view. Mar Ecol Prog Ser 373:203–217. doi:10.3354/meps07768
Pörtner H-O, Farrell AP (2004) Physiology and climate change. Science 322:690–692. doi:10.1126/science.1163156
R Development Core Team (2008) R: a language and environment for statistical computing R Foundation for Statistical Computing, Vienna, Austria ISBN 3-900051-07-0, URL http://www.R-project.org
Riebesell U, Zondervan I, Rost B, Tortell PD, Zeebe RE, Morel FMM (2000) Reduced calcification of marine plankton in response to increased atmospheric CO2. Nature 407:364–367
Ries JB, Cohen AL, McCorkle DC (2009) Marine calcifiers exhibit mixed responses to CO2-induced ocean acidification. Geology 37:1131–1134. doi:10.1130/G30210A.1
Rodhouse PG (1998) Physiological progenesis in cephalopod molluscs. Biol Bull 195:17–20
Rodolfo-Metalpa R, Lombardi C, Cocito S, Hall-Spencer JM, Gambi MC (2010) Effects of ocean acidification and high temperatures on the bryozoan Myriapora truncata at natural CO2 vents. Marine Ecol 31(3):447–456. doi:10.1111/j.1439-0485.2009.00354.x
Rodolfo-Metalpa R, Houlbrèque F, Tambutté É et al (2011) Coral and mollusc resistance to ocean acidification adversely affected by warming. Nature Clim Change 1:308–312
Rodriguez y Baena A, Metian M, Teyssié JL, Debroyer C, Warnau M (2006) Experimental evidence for 234Th bioaccumulation in three Antarctic crustaceans: potential implications for particle flux studies. Marine Chem 100:354–365. doi:10.1016/j.marchem.2005.10.022
Rosa R, Seibel BA (2008) Synergistic effects of climate-related variables suggest future physiological impairment in a top oceanic predator. P Natl Acad Sci USA 105:20776–20780. doi:10.1073/pnas.0806886105
Seibel BA, Fabry VJ (2003) Marine biotic response to elevated carbon dioxide. Adv Appl Biodivers Sci 4:59–67
Seibel BA, Walsh PJ (2001) Potential impacts of CO2 injection on deep-sea biota. Science 294:319–320
Seibel BA, Walsh PJ (2003) Biological impacts of deep-sea carbon dioxide injection inferred from indices of physiological performance. J Exp Biol 206:641–650. doi:10.1242/jeb.00141
Sherrard K (2000) Cuttlebone morphology limits habitat depth in eleven species of Sepia (Cephalopoda: Sepiidae). Biol Bull 198:404–414
Shiao J-C, Lin L-Y, Horng J-L, Hwang P–P, Kaneko T (2005) How can teleostean inner ear hair cells maintain the proper association with the accreting otolith? J Comp Neurol 488:331–341
Spero HJ, Bijma J, Lea DW, Bemis BE (1997) Effect of seawater carbonate concentration on foraminiferal carbon and oxygen isotopes. Nature 390:497–500. doi:10.1038/37333
Stumpp M, Wren J, Melzner F, Thorndyke MC, Dupont S (2011) CO2 induced seawater acidification impacts sea urchin larval development I: elevated metabolic rates decrease scope for growth and induce developmental delay. Comp Biochem Phys A 160(3):331–340. doi:10.1016/j.cbpa.2011.06.022
Stumpp M, Trübenbach K, Brennecke D, Hu MY, Melzner F (2012) Resource allocation and extracellular acid-base status in the sea urchin Strongylocentrotus droebachiensis in response to CO2 induced seawater acidification. Aquat Toxicol 110–111:194–207. doi:10.1016/j.aquatox.2011.12.020
Thomsen J, Melzner F (2010) Moderate seawater acidification does not elicit long-term metabolic depression in the blue mussel Mytilus edulis. Mar Biol 157:2667–2676. doi:10.1007/s00227-010-1527-0
Thomsen J, Gutowska MA, Saphörster J, Heinemann A, Trübenbach K, Fietzke J, Hiebenthal C, Eisenhauer A, Körtzinger A, Wahl M, Melzner F (2010) Calcifying invertebrates succeed in a naturally CO2-rich coastal habitat but are threatened by high levels of future acidification. Biogeosciences 7:3879–3891
Touratier F, Goyet C (2011) Impact of the Eastern Mediterranean Transient on the distribution of anthropogenic CO2 and first estimate of acidification for the Mediterranean Sea. Deep-Sea Res PT I 58:1–15
Tunnicliffe V, Davies KTA, Butterfield DA, Embley RW, Rose JM, Chadwick WW Jr (2009) Survival of mussels in extremely acidic waters on a submarine volcano. Nature Geosci 2:344–348. doi:10.1038/ngeo500
Venn A, Tambutté É, Holcomb M, Allemand D, Tambutté S (2011) Live tissue imaging shows reef corals elevate pH under their calcifying tissue relative to seawater. PLoS ONE 6(5):e20013. doi:10.1371/journal.pone.0020013
Villanueva R, Bustamante P (2006) Composition in essential and non-essential elements of early stages of cephalopods and dietary effects on the elemental profiles of Octopus vulgaris paralarvae. Aquaculture 261:225–240. doi:10.1016/j.aquaculture.2006.07.006
Waldbusser GG, Bergschneider H, Green MA (2010) Size-dependent pH effect on calcification in post-larval hard clam Mercenaria spp. Mar Ecol Prog Ser 417:171–182
Weiss IM, Tuross N, Addadi L, Weiner S (2002) Mollusc larval shell formation: amorphous calcium carbonate is a precursor phase for aragonite. J Exp Zool 293:478–491. doi:10.1002/jez.90004
Wood HL, Spicer JI, Widdicombe S (2008) Ocean acidification may increase calcification rates, but at a cost. Proc Roy Soc B 275:1767–1773. doi:10.1098/rspb.2008.0343
Zondervan I, Zeebe RE, Rost B, Riebesell U (2001) Decreasing marine biogenic calcification: a negative feedback on rising atmospheric pCO2. Global Biogeochem Cy 15:507–516
Acknowledgments
Authors thank the reviewers and the editor of Marine Biology for their comments that strongly improved this paper. The International Atomic Energy Agency is grateful to the Government of the Principality of Monaco for the support provided to its Environment Laboratories. This work was supported by the IAEA. This work is a contribution to the “European Project on Ocean Acidification” (EPOCA) which received funding from the European Community’s Seventh Framework Programme (FP7/2007-2013) under grant agreement no. 211384. Ms. André and Eric Rinaldi of Monaco are thanked for the collection of cuttlefish specimens. Mr. Antoine and Mrs. José Lacoue-Labarthe are thanked for providing juveniles of crabs and shrimps.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by S. Dupont.
Rights and permissions
About this article
Cite this article
Dorey, N., Melzner, F., Martin, S. et al. Ocean acidification and temperature rise: effects on calcification during early development of the cuttlefish Sepia officinalis . Mar Biol 160, 2007–2022 (2013). https://doi.org/10.1007/s00227-012-2059-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-012-2059-6